Fungalpedia – Note 197, Crepidotus
Crepidotus (Fr.) Staude
Citation if using this entry: Bera et al. 2024 (in prep) – Fungalpedia, Basidiomycota 1.
Index Fungorum, MycoBank, Facesoffungi, GenBank, Fig. 1
Classification: Crepidotaceae, Agaricales, Agaricomycetes, Basidiomycota, Fungi
Fries (1821) initially categorized Crepidotus under Agaricus trib. Crepidotus Fr., which included pleurotoid taxa with fibrillose veil and pale or ferruginous basidiospores (Senn-Irlet 1995). However, Crepidotus was later raised to the generic rank by Staude (1857) (Donk 1949, Consiglio & Setti 2008, Na et al. 2022). The tribe Crepidotus used to encompass eight species, most of which have been transferred to two current genera: Entoloma (Fr.) P. Kumm. and Tapinella E.-J. Gilbert E.-J. Gilbert, leaving only three species that persisted within Crepidotus (Consiglio & Setti 2008, Na et al. 2022). The first contributions towards the diversity of this genus in the western hemisphere were by Peck (1886) and Murrill (1916, 1917). The comprehensive revisionary work by Hesler & Smith (1965) provided more detailed documentation of the North American species of Crepidotus. Subsequently, the diversity of this genus has also been studied in Asian countries, resulting in the discovery of many novel species (Na et al. 2022). Members of Crepidotus have typical pleurotoid basidiomata with a rudimentary stipe and dark-coloured basidiospores (Guzmán-Dávalos et al. 2017, Capelari 2011, Kumar et al. 2020, Na et al. 2022). Various shapes can be found in the basidiomata, including semicircular, flabelliform, reniform, or spathuliform (Senn-Irlet 1995). The lamellae spacing is relatively uniform and can be subventricose to rarely ventricose (Senn-Irlet 1995). The colour of the spore deposit has been regarded as one of the defining characteristics of Crepidotus (Senn-Irlet 1995).
Another unique characteristic of Crepidotus is the absence of pleurocystidia but the presence of cheilocystidia (Kumar et al. 2018). The basidiospores of Crepidotus can be categorized into two types: smooth and ornamented (Na et al. 2022). According to the classification of Consiglio & Setti (2008), the ornamented basidiospores are generally associated with clamped hyphae, whereas the smooth basidiospores mostly occur with hyphae without any clamp (Senn-Irlet 1995, Consiglio & Setti 2008, Na et al. 2022). Even if the distinct morphological characteristics could define the species of this genus, molecular studies have proven to be quite advantageous in species delimitation (Jančovičová et al. 2014, Kasuya et al. 2014, Ge et al. 2017, Guzmán-Dávalos et al. 2017). This saprotrophic genus has a worldwide distribution, mostly on dead tree logs, and approximately 200 species have been documented under Crepidotus (Senn-Irlet 1995, Consiglio & Setti 2008, Kirk et al. 2008, Na et al. 2022).
Synonyms: Agaricus trib. Crepidotus Fr., Calathinus Quél., Cyphellathelia Jülich, Dochmiopus Pat., Octojuga Fayod, Phaeoglabrotricha W.B. Cooke, Phaeomyces E. Horak, Phialocybe P. Karst., Pleurotellus Fayod, Tremellastrum Clem., Tremellopsis Pat.
Type species: Crepidotus mollis (Schaeff.) Staude
Other accepted species: (Species Fungorum – search Crepidotus)
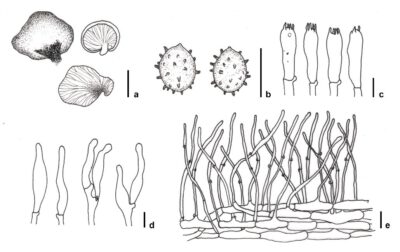
Figure 1 – Crepidotus yuanchui (FFAAS 0339). a Basidiomata. b Basidiospores. c Basidia. d Cheilocystidia. e Pileipellis. Scale bars: a = 1 cm, b, d & e = 10 μm, c = 5 μm. Redrawn from Na et al. (2022).
References
Fries EM. 1821 – Systema mycologicum. Vol. 1. Sweden, Lundin, 520 pp.
Murrill WA. 1916 – Agaricaceae. Agariceae. North American Flora, 9, 1–542.
Murrill WA. 1917 – Agaricaceae. Agariceae. North American Flora, 10, 145–226.
Peck CH. 1886 – New York species of Pleurotus, Claudopus and Crepidotus. Annual Report of the Trustees of the State Museum of Natural History, 39, 58–73.
Entry by
Ishika Bera, Center of Excellence in Fungal Research, Mae Fah Luang University, Chiang Rai 57100, Thailand
(Edited by Kevin D Hyde, Samaneh Chaharmiri-Dokhaharani, & Achala R. Rathnayaka)
Published online 6 February 2024